If you work in a field that requires gases, it’s important to know how to find partial pressure. partial pressure exerted by an individual gas in a mixture of gases. it can be measured using a variety of methods.
So In this blog post, we’ll discuss how to find partial pressure and why it’s important when dealing with gases. We’ll also look at some examples to help illustrate the concept. By the end of this article, you should have a better understanding of finding partial pressures. and why it’s so important.
Unveiling the secrets of gas mixtures! Learn how to find the partial pressure of a specific gas using concepts like Dalton’s Law, mole fraction, and the ideal gas law. Whether you have the total pressure and mole fraction, or just moles and want to find the partial pressure using Boyle’s, Charles’s, or Avogadro’s Law, this exploration will equip you to handle gas mixtures like a pro!
Contents
- 1 1. The Best Way to Find Partial Pressure Without Volume
- 2 2. The Calculation Method to Find Partial Pressure
- 3 3. Partial Pressure From
- 4 4. Find Partial Pressure in mmHg
- 5 5. Partial pressure from mole Formula
- 6 6. Total partial pressure formula
- 7 7. Method of execution of the partial formula
- 8 Conclusion
1. The Best Way to Find Partial Pressure Without Volume
It is possible to find partial pressure without knowing the volume of a gas, as long as you know the total pressure and the mole fraction of the gas in the mixture. The mole fraction is the ratio of the number of moles of the gas to the total number of moles of all the gases in the mixture.
The partial pressure of a gas is equal to the total pressure multiplied by its mole fraction. So if you know the total pressure and the mole fraction of the gas in the mixture, you can calculate and find partial pressure using the following formula:
Partial pressure = Total pressure x Mole fraction
For example, if you have a mixture of gases with a total pressure of 1 atm, and the mole fraction of gas A is 0.2, then the partial pressure of gas A would be:
Partial pressure of gas A = 1 atm x 0.2 = 0.2 atm
2. The Calculation Method to Find Partial Pressure
You must first determine the number of moles of a gas present to determine its partial pressure. The Ideal Gas Law can be applied to accomplish this: PV equals nRT, where R is the universal gas constant, T is the temperature, n is the number of moles, and P is pressure. You can use the Ideal Gas Law once more to solve for partial pressure after determining the moles of gas. The mole fraction times the total pressure is equal to the partial pressure.
Curious about the pressure of a gas in a mixture? While fancy tools like gas chromatography or mass spectrometry can analyze the components, for ideal gas mixtures, knowing the equilibrium constant (Kp) can help you find the partial pressure of each gas using Raoult’s law! Discovering partial pressure involves understanding fundamental science laws. These laws govern various phenomena in nature and play a crucial role in calculating partial pressures in different environments.
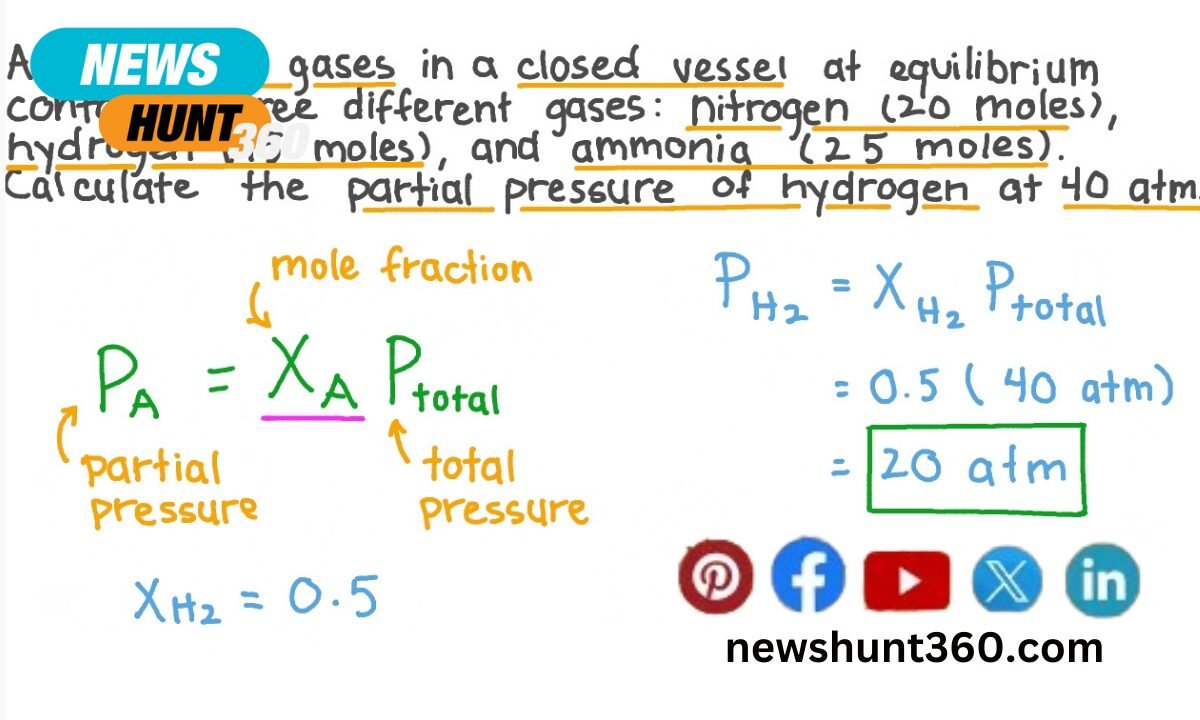
3. Partial Pressure From
To find the partial pressure of gas from the amount of gas in grams. After you will need to use the molar mass of the gas. The molar mass is the number of grams per mole of the gas. Then you have the molar mass. And you can use the Ideal Gas Law to find the partial pressure. In NEET Chemistry Preparation, understanding how to find partial pressure is essential for mastering gas laws and equilibrium concepts.
4. Find Partial Pressure in mmHg
To find the partial pressure of a gas, you will need to use the Ideal Gas Law. This law states that:
PV = nRT
where P is the pressure of the gas, V is the volume of the gas, n is the number of moles of the gas, R is the universal gas constant, and T is the temperature.
To find the partial pressure of a gas, you will need to rearrange this equation to solve for P. Once you have done that, plug in the values for V, n, R, and T. The units for P will be in mmHg (millimeters of mercury).
5. Partial pressure from mole Formula
The easiest way to find the partial pressure of a gas is to use the Ideal Gas Law. This law states that PV=nRT, where P is the pressure, V is the volume, n is the number of moles, R is the universal gas constant, and T is the temperature. To solve for partial pressure, you will need to know two of these variables. For example, if you know that the temperature is 25 degrees Celsius and the volume is 2 liters, you can plug those values into the equation to solve for P.
6. Total partial pressure formula
The total partial pressure formula i is applied to calculate the sum of the partial pressures of all the gases in a mixture. This is very important to know because it helps us to deeply study and determine the gas mixture’s properties. Such as its density and viscosity You must understand the partial pressures of all the gases in the mixture as well as its application. Moreover, the partial pressure of a gas is its contribution to the overall pressure of the mixture. It is usually expressed in units of the atmosphere (atm).
Once you have the partial pressures of all the gases, you can add them together using the following equation:
Ptotal = P1 + P2 + P3 + …
where P1, P2, and P3 are the partial pressures of each gas in the mixture.
When working with gas mixtures, chemists and other scientists might benefit from the total partial pressure formula. It can assist students in comprehending the effects of the interactions between various gases. and they possess a mixture of general characteristics.
7. Method of execution of the partial formula
The method of execution of the partial formula is as follows:
1) First, the equation is solved for the desired variable.
2) Next, the molar mass of each gas will be calculated.
3) Thirdly mole fractions of each gas are then determined.
4) Finally, the partial pressure of the desired gas will be calculated using the mole fractions and the total pressure.
Conclusion
This is all about gas behavior chemistry. with the help of Dalton’s laws of partial pressure and its execution, an informative guide is here. In the end, You will find out how individual gas mixtures are calculated. For Further updates keep in touch with News Hunt.




